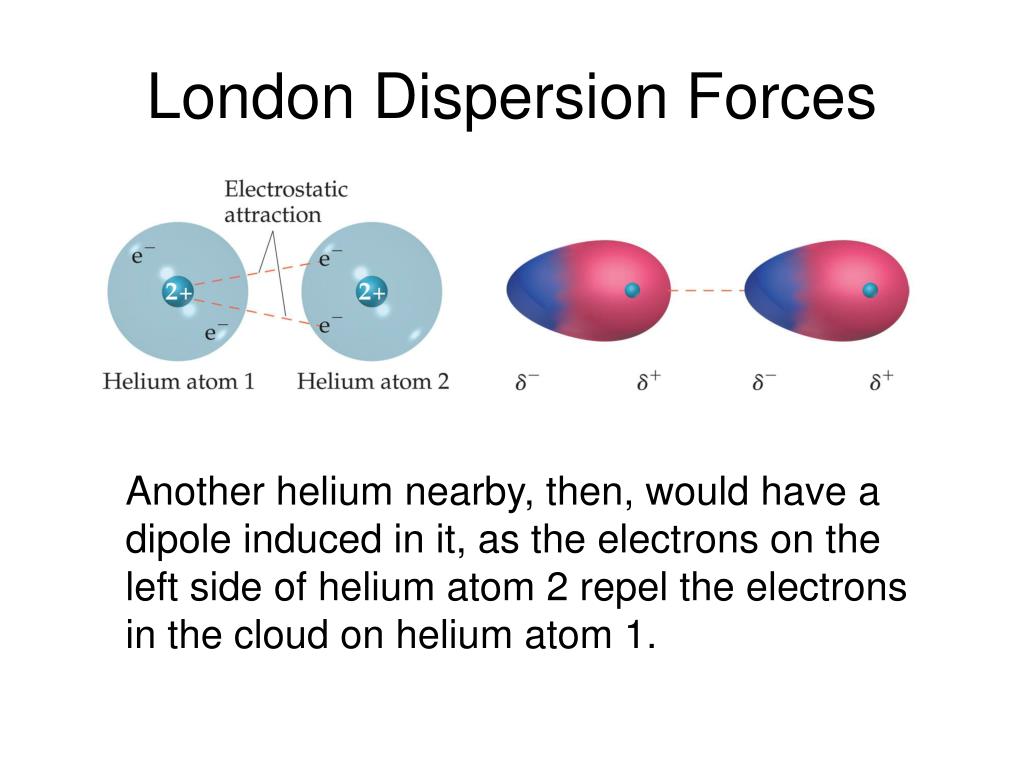
London forces are the attractive forces that cause nonpolar substances to condense to liquids.So i have just finished reading a section of my pchem book.
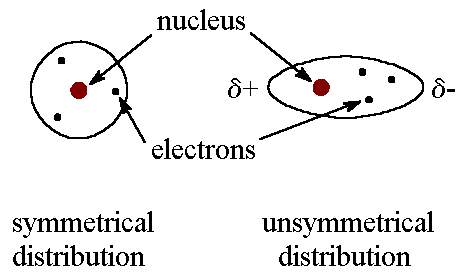
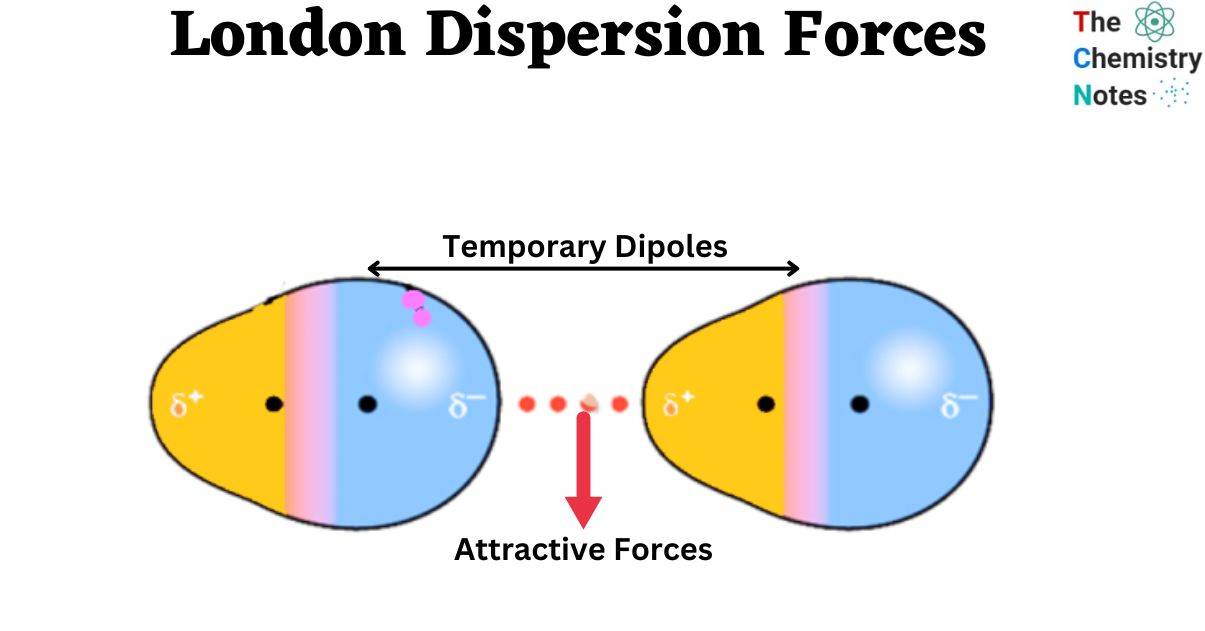
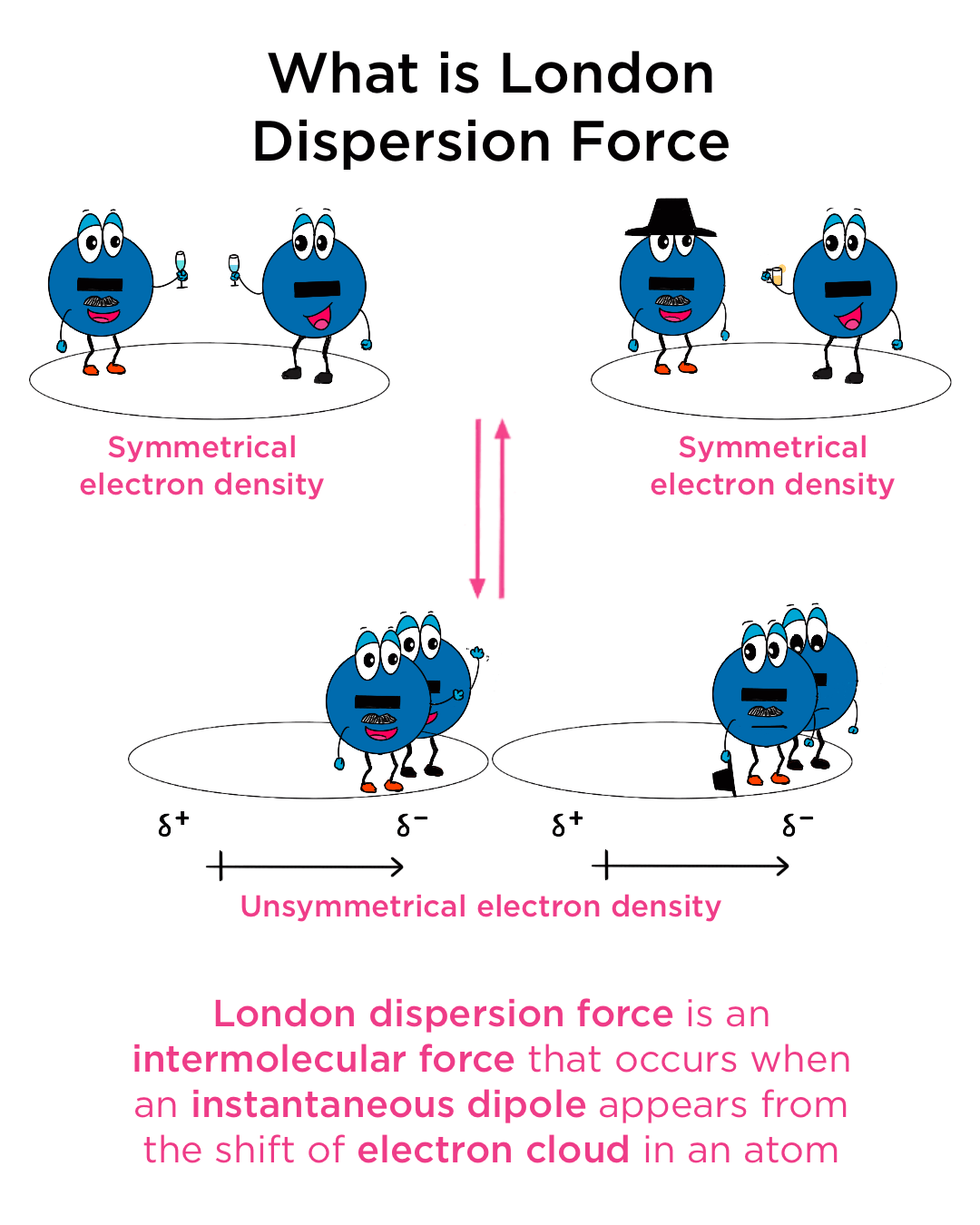
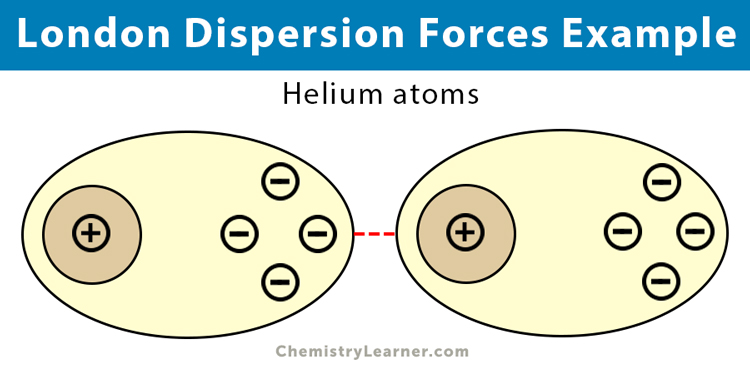
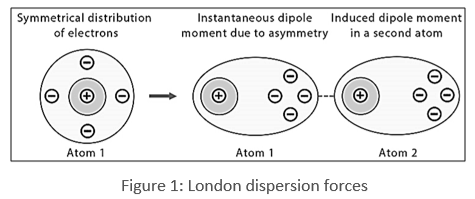
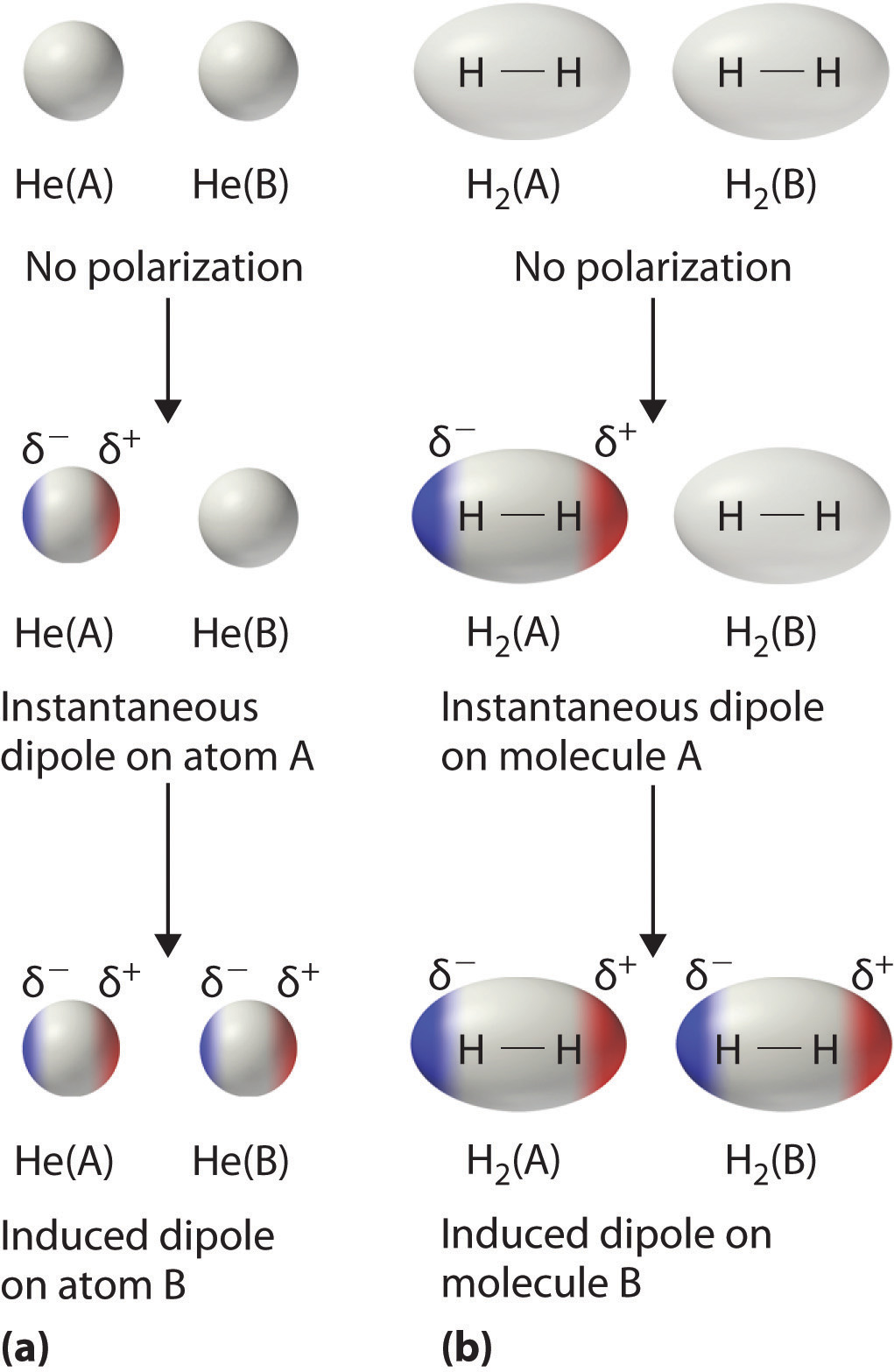
The london dispersion force is the weakest intermolecular force.Because the electrons move around a lot, sometimes they may move in a way that creates a temporary dipole moment.They are temporary attractive forces that result when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles.
The london dispersion force is defined as the weakest intermolecular force.To this end, we have set an ambitious target to achieve net zero for our portfolio by 2040, as well as interim targets to reduce emissions 35% by 2025 and 60% by 2030.
More electrons/greater surface area means more ldf.These forces are weakest among all intermolecular attractions.The london dispersion force is sometimes called a.
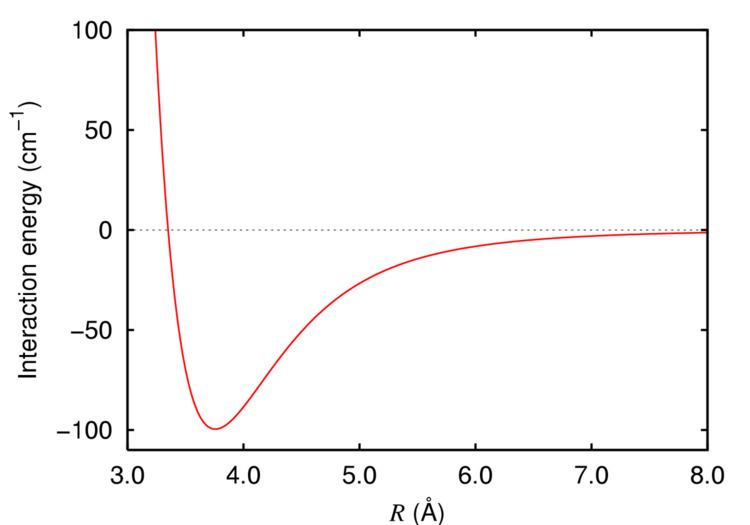
At london civ, we believe we have a responsibility to manage our impact on the world, to help create a safe and stable future for our partner fund beneficiaries, and for everyone.Dispersion forces are present between all molecules (and atoms) and are typically greater for heavier, more polarizable molecules and molecules with larger surface areas.
This is a temporary and appealing force that provides the results if the electrons in two are adjacent as well as they occupy positions that turn the atoms into temporary dipoles.
Last update images today What Is London Dispersion Forces
 NHL Free Agency 2024: Grades For Reinhart, Guentzel, Lindholm
NHL Free Agency 2024: Grades For Reinhart, Guentzel, Lindholm
Lancashire 344 for 8 (Jennings 183, James 3-53) vs Nottinghamshire - no play on day two
James Anderson's preparations for his farewell Test, at Lord's next week, suffered a setback after rain washed out the entire second day of the Vitality County Championship match between Lancashire and Nottinghamshire at Southport.
Despite early showers, umpires Martin Saggers and Tom Lungley initially decreed that Lancashire would resume their first innings at 12.30pm, but prolonged rain returned to scupper that plan and the officials finally pulled the plug on the day's cricket at 4.05pm.
The abandonment was a particular disappointment to spectators who had travelled to Trafalgar Road in the hope of seeing Anderson bowl in his first game of the season, and what might yet be his last game for Lancashire.
He is due to play his 188th and final Test match against West Indies at Lord's, starting on July 10, and though he had previously indicated that he would be keen to continue his county career, Rob Key, England's director of cricket, confirmed on Monday that Anderson is to stay on with the England team after the Lord's Test, to fulfil a new role as a fast-bowling mentor.
Should play be possible on Tuesday, Lancashire will resume on 344 for eight with skipper Keaton Jennings on 188 not out.
In the nine games played at Southport since first-class cricket returned to the ground after a 12-year absence in 2011, this is only the second time in 31 days that play has been completely abandoned.