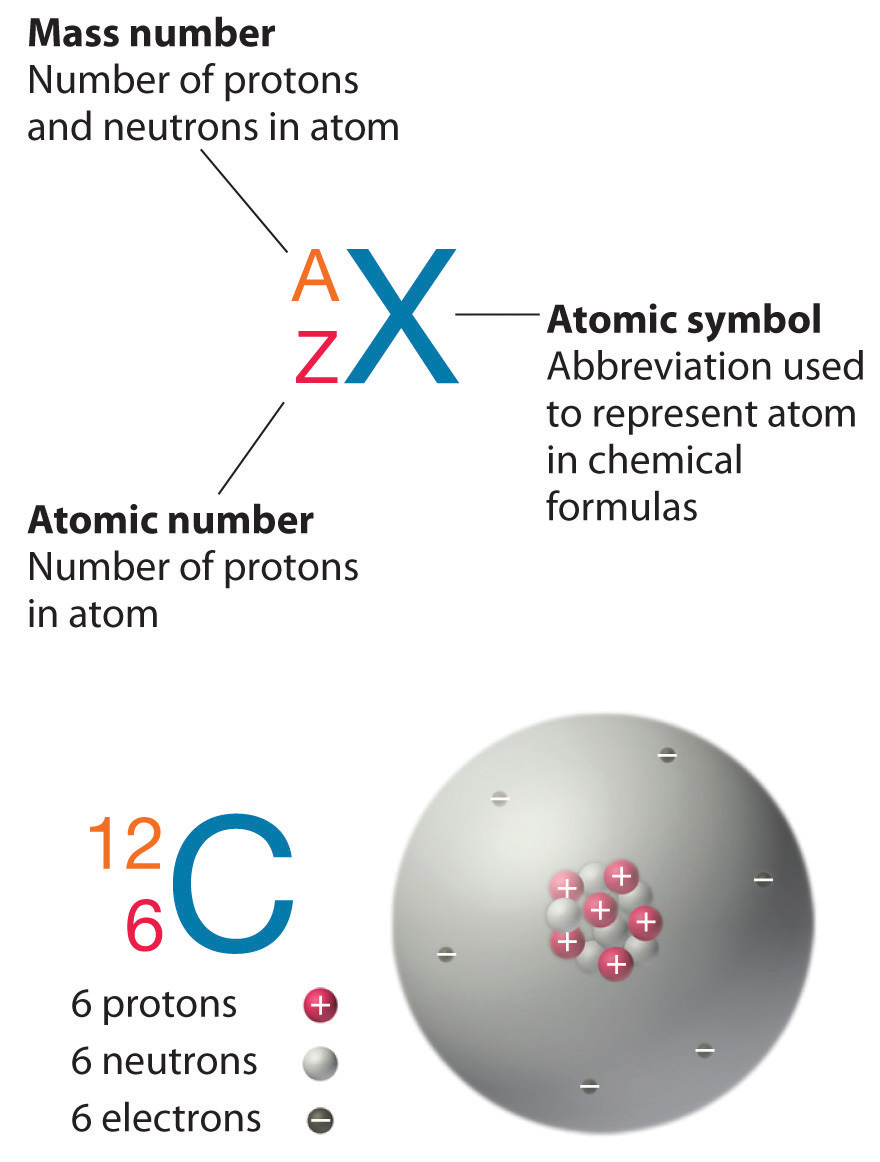Only the stable isotope occurs in nature 133 cs before.132.8752821 u (calculated nuclear mass without electrons) mass excess:
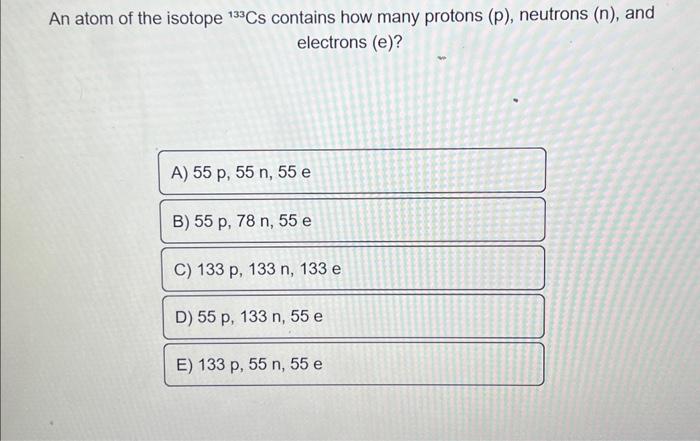
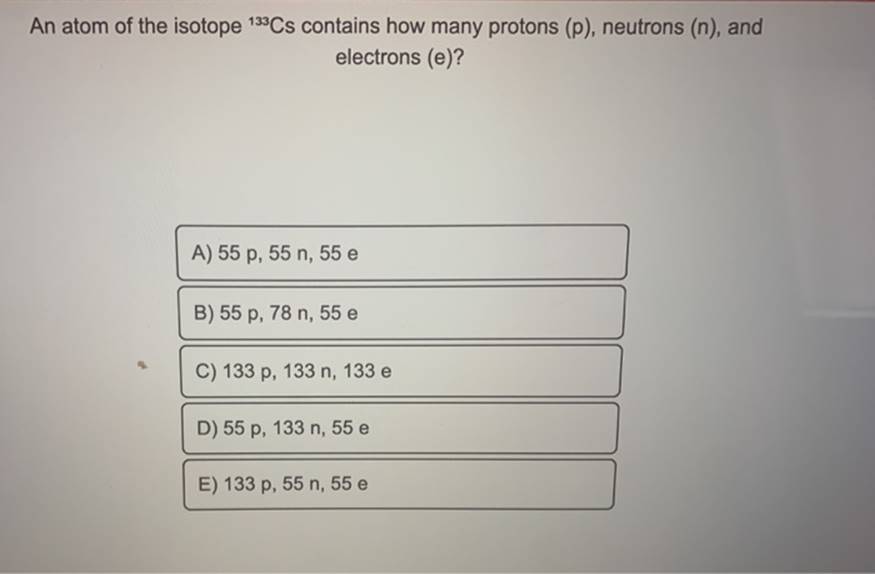
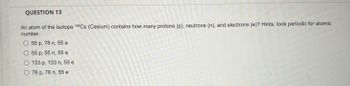
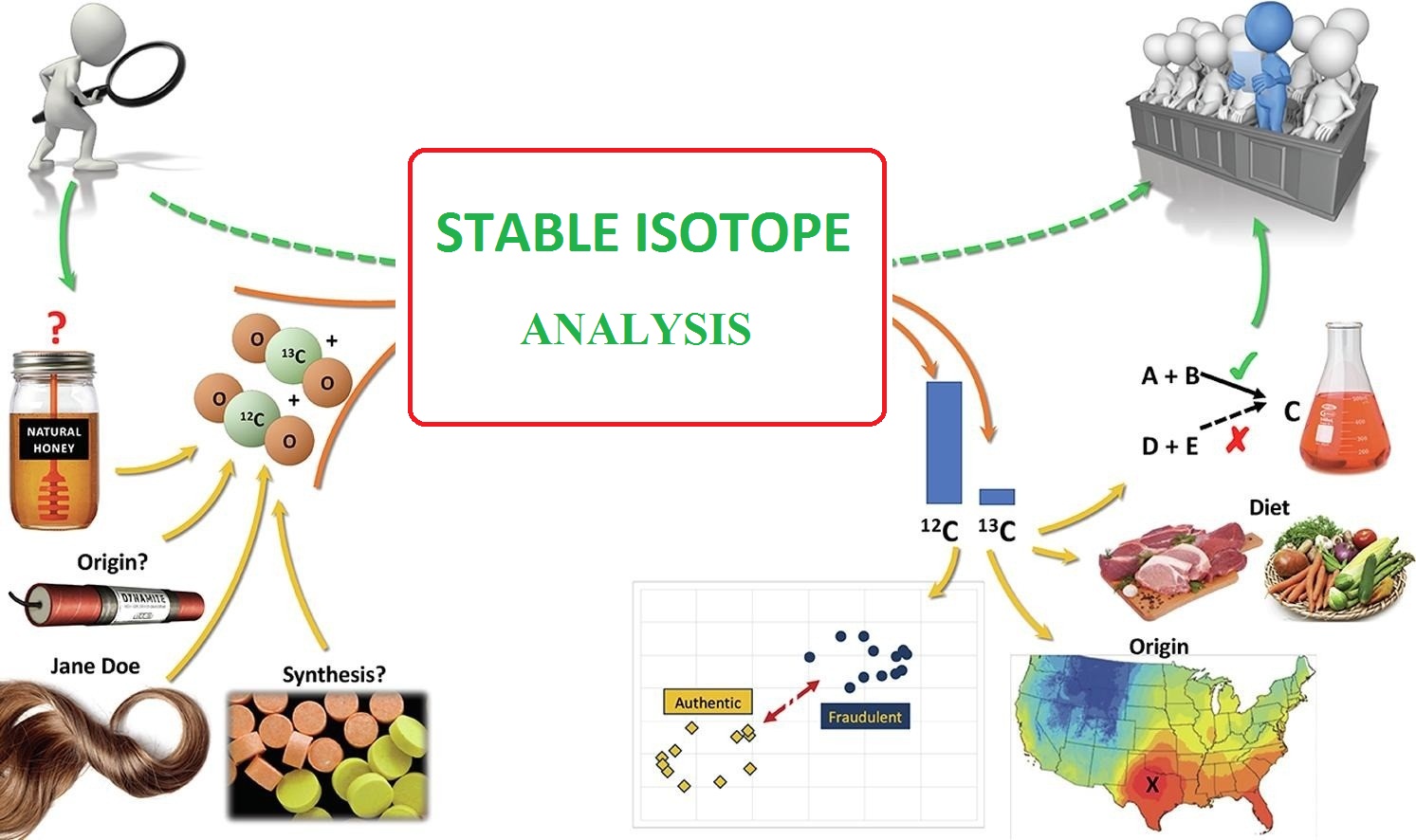
An atom of the isotope 133cs contains how many protons (p), neutrons (n), and electrons (e)?Since the atomic mass of this isotope is 133, and atomic mass = number of protons + number of neutrons.Stable isotope tracing has been in use for years but its application has been limited to looking for specific metabolites of a.
2) know the atomic number (z) of the cesium atom (cs):A) 55 p, 55 n, 55 e b) 55 p, 78 n, 55 e c) 133 p, 133 n, 133 e d) 55 p, 133 n, 55 e e) 133 p, 55 n, 55 e w.
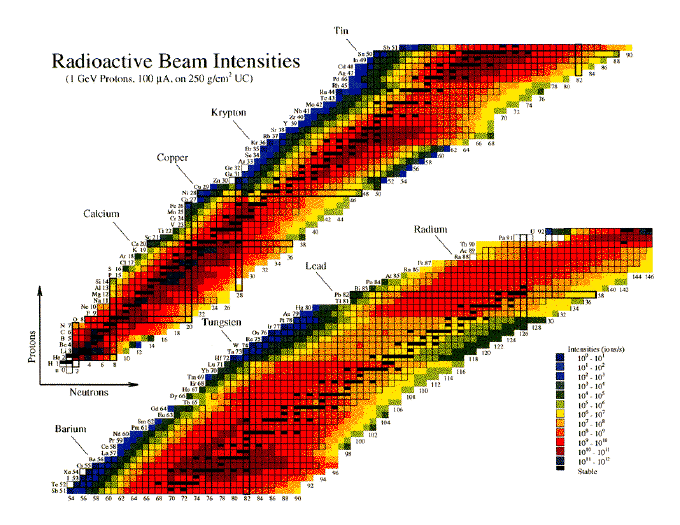
There are 2 steps to solve this one.However, since the chernobyl nuclear disaster in 1986, large parts of the population have found the element cesium in the form of the radioactive fission product 137 cs became known, which came to a large extent in the environment.Caesium (55 cs) has 41 known isotopes, the atomic masses of these isotopes range from 112 to 152.
Unknown element branium has two isotopes.Verified solution this video solution was recommended by our tutors as helpful for the problem above
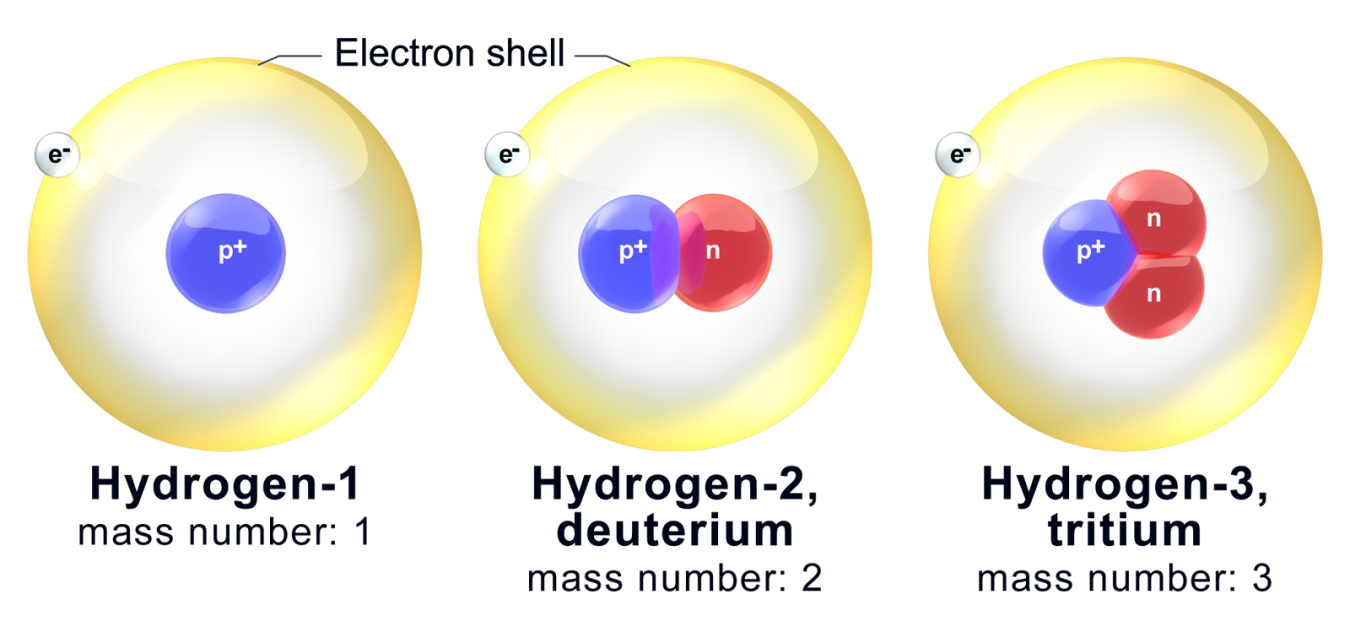
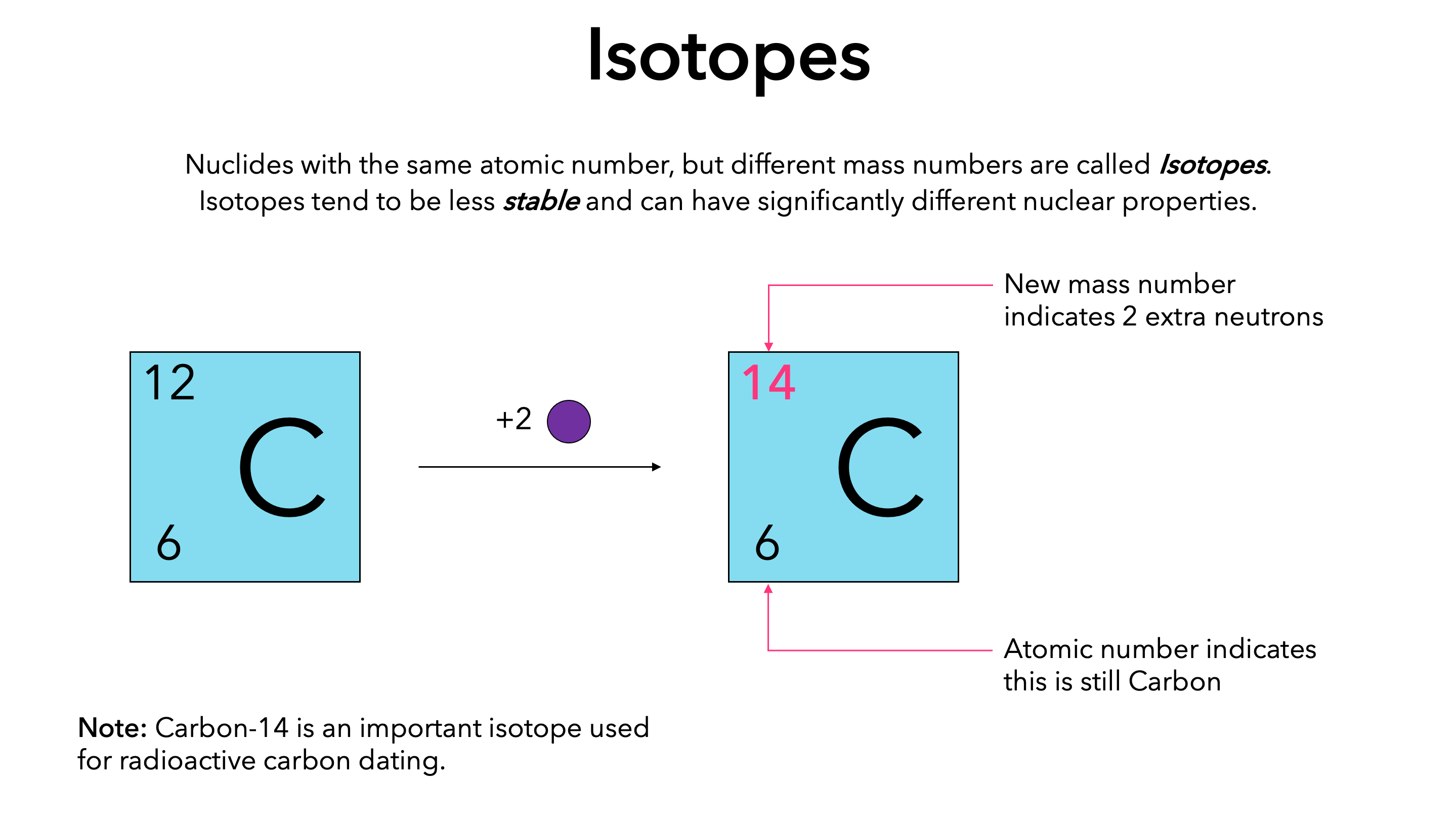
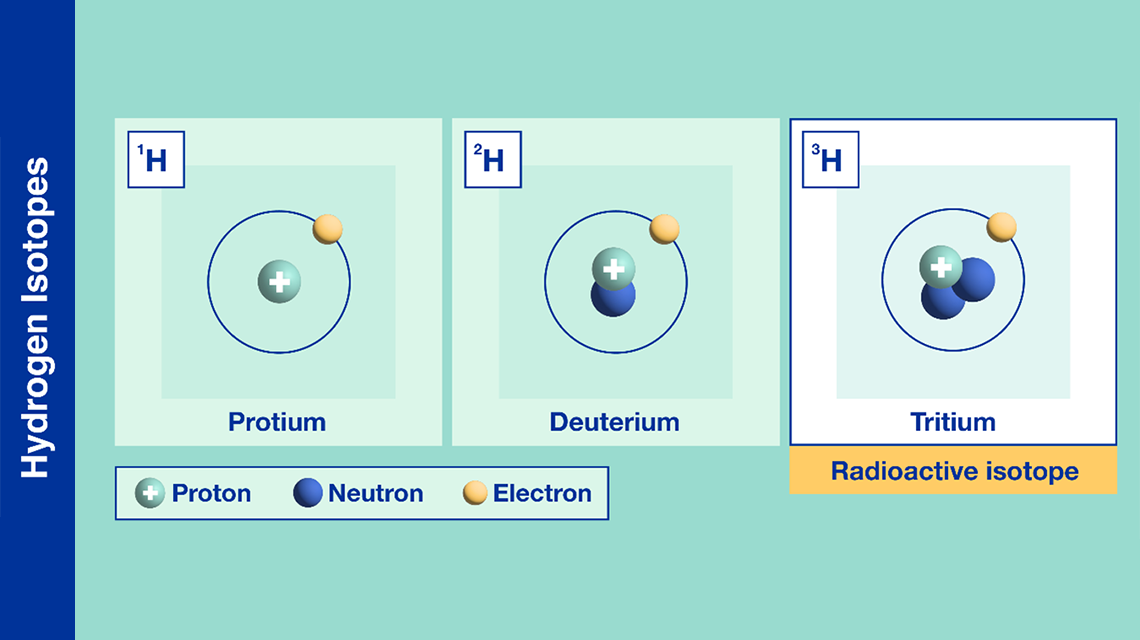
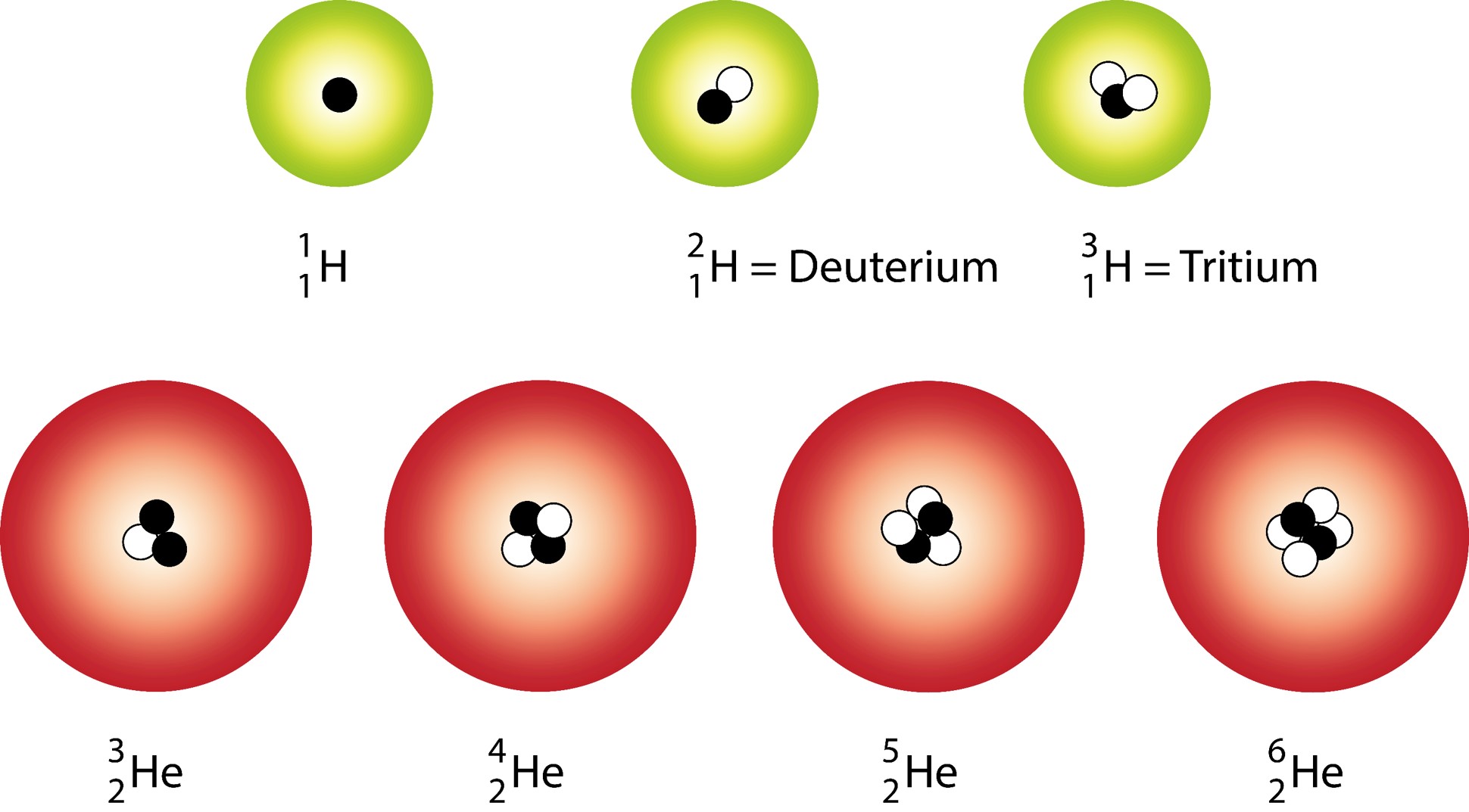
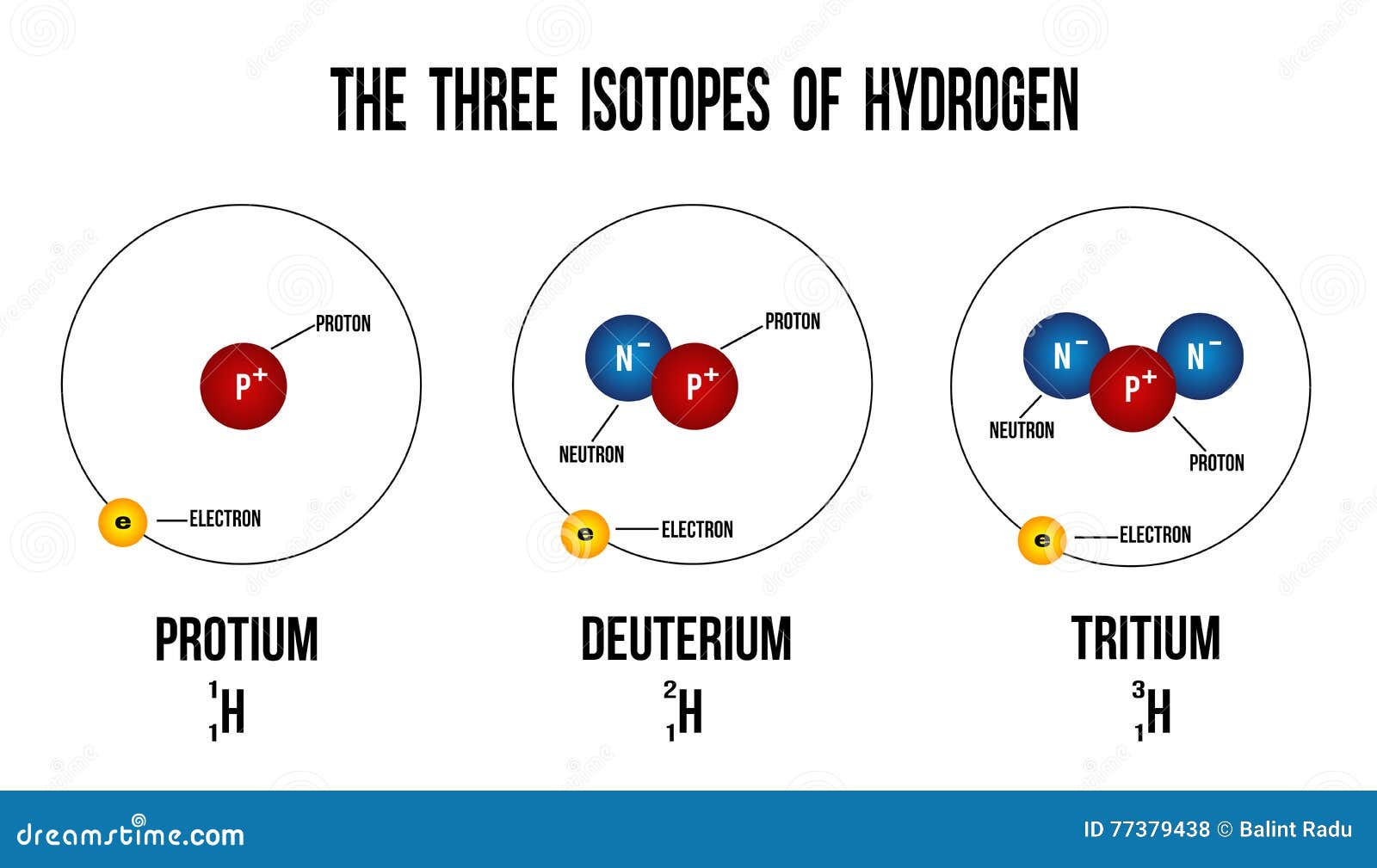
The species that are of the same element but with different numbers of neutrons are called isotopes.Neutrons, electrons, and protons of an isotope.This isotope is stable and thus has no decay products, so instead we show decay chains that lead down to it.
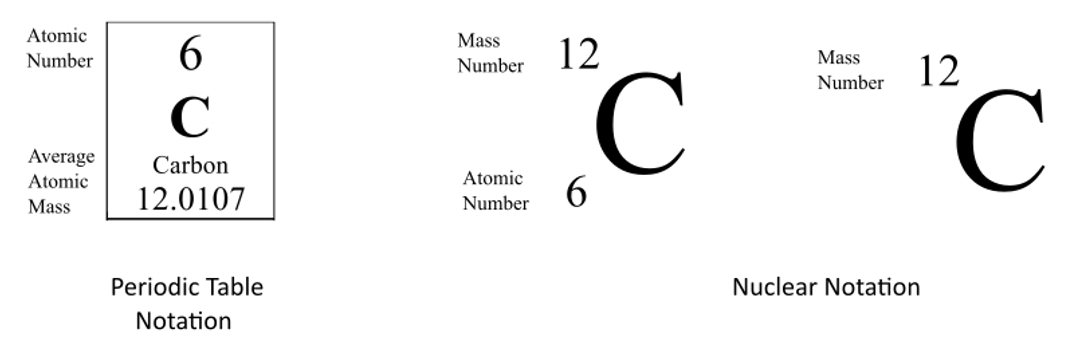
133 cs or 13355 cs mass number a:The atomic masses range from 112 to 148.
133 (= number of nucleons) atomic number z:
Last update images today An Atom Of The Isotope 133cs
 Julio Rodríguez Homers, J.P. Crawford Drives In Three And Mariners Beat Orioles 7-3 To Avoid Sweep
Julio Rodríguez Homers, J.P. Crawford Drives In Three And Mariners Beat Orioles 7-3 To Avoid Sweep
Plan ahead in fantasy baseball with help from our forecaster projections. Each day, we will provide an updated preview of the next 10 days for every team, projecting the matchup quality for hitters (overall and by handedness) as well as for base stealers.
This page will be updated daily throughout the season, so be sure to check back often for the latest 10-day outlook.
For our 10-day projections for each team's pitching matchups, click here.












/alpha-decay-d1cdcfc8400c43798b9930002b0e96cb.jpg)